|
The molecule H 2 is composed of two H atoms. 
Total number of molecular orbitals is equal to the total number of atomic orbitals used to make them. The principles to apply when forming pictorial molecular orbitals from atomic orbitals are summarized in the table below: Principle Once you have the molecular orbitals and their energy ordering the ground state configuration is found by applying the Pauli principle, the aufbau principle and Hund's rule just as with atoms. Each molecular orbital can only have 2 electrons, each with an opposite spin. Similar to atomic orbitals, molecular orbitals are wave functions giving the probability of finding an electron in certain regions of a molecule. In molecules, atomic orbitals combine to form molecular orbitals which surround the molecule. "Third-order perturbation theory for van der Waals interaction coefficients" (PDF). ^ Tang, Li-Yan Yan, Zong-Chao Shi, Ting-Yun Mitroy, J."The Li 2 F "shelf" state: Accurate potential energy curve based on the inverted perturbation approach". "New observation of the, 13Δg, and 23Πg states and molecular constants with all 6Li 2, 7Li 2, and 6Li 7Li data". "Potential energy Lambda double and Born-Oppenheimer breakdown functions for the B 1Pi u "barrier" state of Li 2". "High-resolution photoassociation spectroscopy of the 6Li 2 c-state". Madison, High resolution photoassociation spectroscopy of the 6Li 2 A-state, 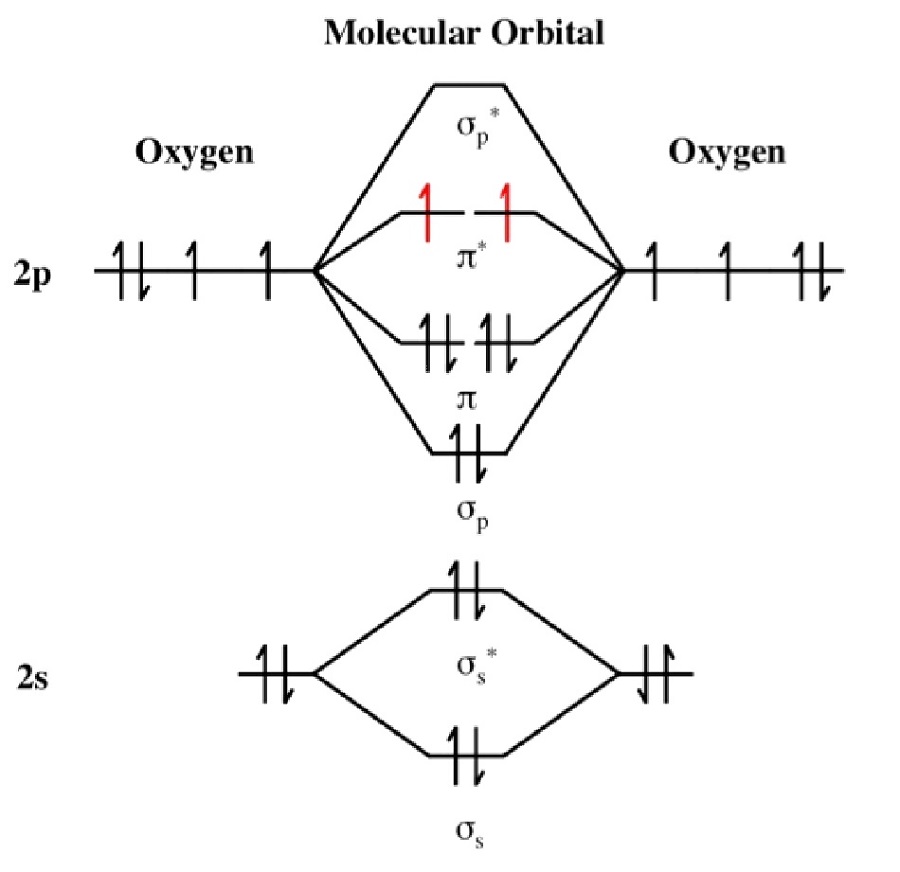
"A DPF data analysis yields accurate analytic potentials for Li 2(a)and Li 2(c) that incorporate 3-state mixing near the c-state asymptote". "Accurate analytic potentials for Li 2(X) and Li 2(A) from 2 to 90 Angstroms, and the radiative lifetime of Li(2p)". ^ a b c d e f g h i j k Le Roy, Robert J.This lithium oscillator strength is related to the radiative lifetime of atomic lithium and is used as a benchmark for atomic clocks and measurements of fundamental constants. in is more precise than any previously measured atomic oscillator strength. For example, the C 3 value for atomic lithium extracted from the A-state potential of Li 2 by Le Roy et al. 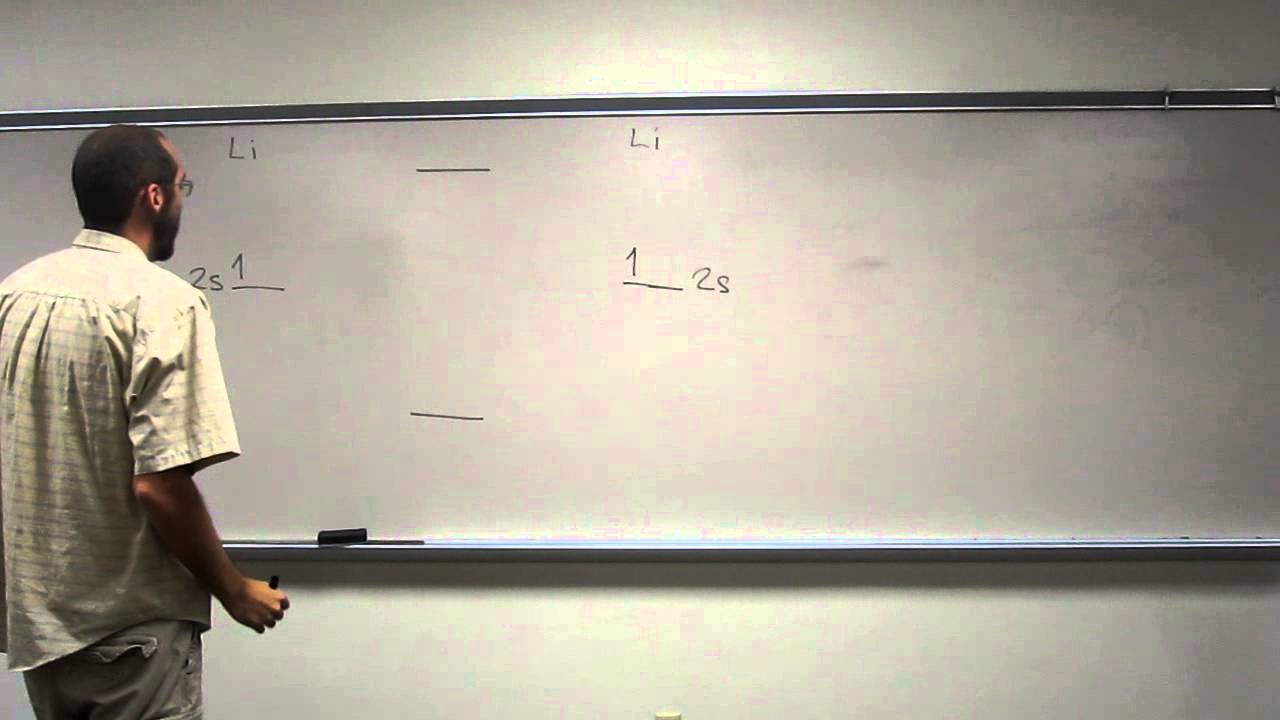
Li 2 potentials are often used to extract atomic properties. The most reliable of these potential energy curves are of the Morse/Long-range variety (see entries in the table below). Analytic empirical potential energy curves have been constructed for the X-state, a-state, A-state, c-state, B-state, 2d-state, l-state, E-state, and the F-state. It is the most thoroughly characterized compound in terms of the accuracy and completeness of the empirical potential energy curves of its electronic states. īeing the lightest stable neutral homonuclear diatomic molecule after H 2, and the helium dimer, dilithium is an extremely important model system for studying fundamentals of physics, chemistry, and electronic structure theory. It has been observed that 1% (by mass) of lithium in the vapor phase is in the form of dilithium. The electron configuration of Li 2 may be written as σ 2. It has a bond order of 1, an internuclear separation of 267.3 pm and a bond energy of 102 kJ/mol or 1.06 eV in each bond. Dilithium, Li 2, is a strongly electrophilic, diatomic molecule comprising two lithium atoms covalently bonded together.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
